Book Review
Discoveries of a Non-Reductionist
by Liona Fan Chiang
October 2011
A PDF of this article, published in the October 16, 2015 issue of Executive Intelligence Review, is re-published here with permission.
Cosmophysical Factors in Stochastic Processes
by Simon E. Shnoll
Rehoboth, New Mexico: American Research Press, 2012
Translated by Alexey V. Agafonov and Olga Seraya from the original Russian edition of 2009, 433 pages, available online at: http://shnoll.ptep-online.com/publications/shnoll2012.pdf
Oct. 11The journey of Simon Elevich Shnolltoday a professor of physics at Moscow State Universitystarted at the beginning of the atomic age in 1951, when he was employed to prepare radioactive solutions in a branch of the Atomic Project, the newly reorganized Department of Medical Radiology at the Central Institute for Physician Excellence in Moscow. On the side, at the end of the workday, he performed experiments in his specialty field, biochemistry.
ATP and ATPase:
|
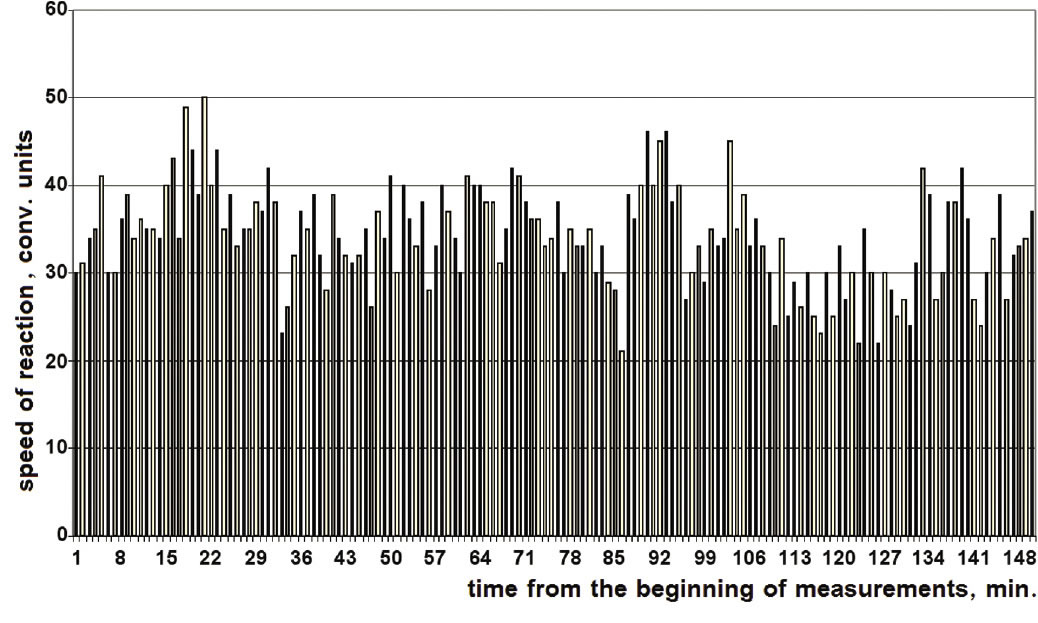
He began with experiments measuring enzymatic (ATPase) activitythe use of ATP by the proteins of the actomyosin complex, a complex that controls muscle contractionand noticed something strange. The reaction rate seemed to change dramatically over short periods of time. Having taken a measurement of reaction rates every 15 seconds, he found that the rate of reaction did not fluctuate about a particular average rate, but rather varied wildly, sometimes smoothing out and sometimes jumping by a factor of two.
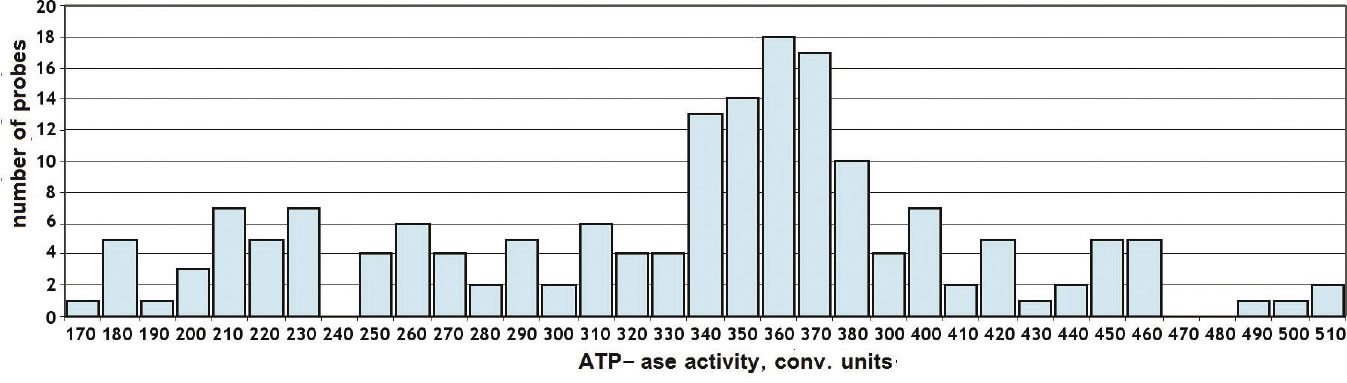
The amount of variation of the reaction rate seemed sporadic, but it was not random. The rates were discrete. When the rates themselves were tracked, it could be seen that there were preferred values. There were also, anomalously, rates which never occurred; they were forbidden. In an attempt to see if the discreteness would disappear, he eventually increased the number of measurements to hundreds, yet, the discreteness would not disappear; instead it became more distinct. (Figure 1) [Click on any figure to view full size. ]
FIGURE 2
Shnoll 2012
Original Caption: Figure 1-2. The histogram distribution of realized values of the experiment depicted in Fig. 1-1 [omitted], conducted on October 5, 1957. |
Figure 2 shows a bar graph (histogram) of these rates. It tabulates the number of times each reaction rate occurred. As can be seen in the graph, the reaction rate of 360 units of ATPase activity per unit of time occurred most frequently, while the rate of 240 units was never registered.
It took several more experiments to prove that the recorded changes in enzyme activity were due to changes in the state, or conformation, of many proteins on a macroscale. In other words, though the proteins are expected to exist in several almost equiprobable states, and individual molecules change conformation rather quickly (10-7 seconds), yet somehow a form of synchronization was occurring, manifested as a macroscopic effect (the rate of activity). They were synchronous in macrovolume, according to Shnoll. (Figure 3)
Persistent Synchronicity
FIGURE 3
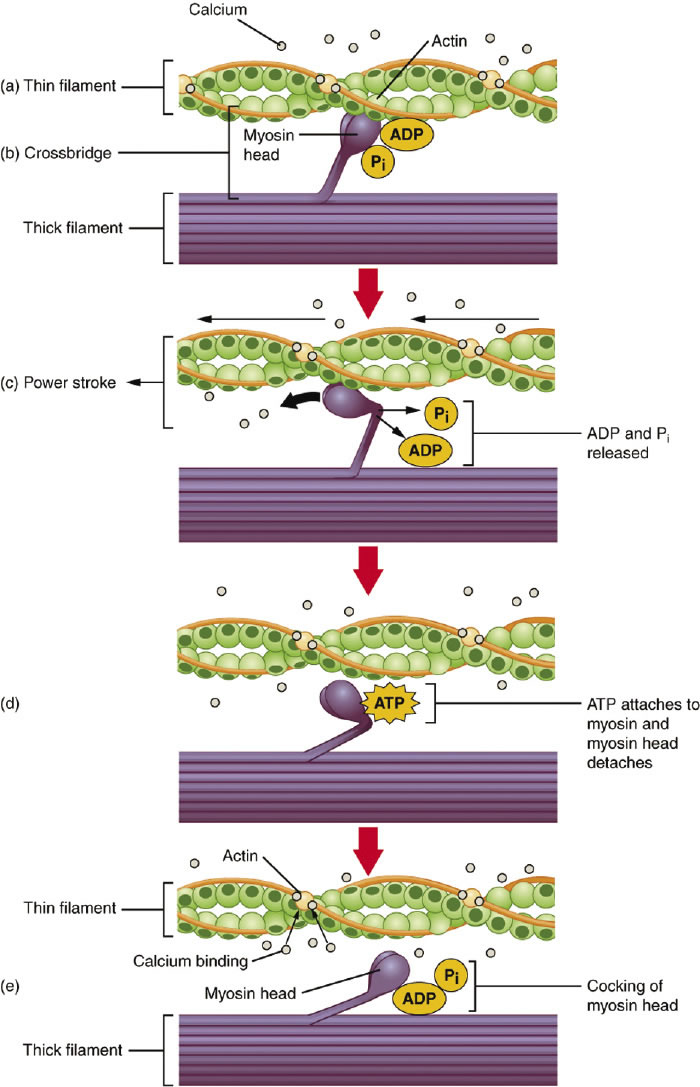
Anatomy & Physiology, an OpenStax College textbook,
http://cnx.org/content/col11496/1.6/, June 19, 2013, unit 2-6-4. States of conformation in the actin myosin complex in skeletal muscle contraction. ADP is adenosine diphosphate; Pi is inorganic phosphate. |
In an attempt to narrow down possible causes and factors of influence, Shnoll took measurements from various parts of the volume. He found the same changes in reaction rate. He even took a common solution and poured it into several vials and measured those at the same time. The conformation changes were still synchronized. That is, though the rates continued to fluctuate, seemingly erratically, those fluctuations occurred at the same time for each separate sample.
He tested this repeatedly, spending years trying to detect all possible errors: fluctuations in mechanics, temperature, pressure, and so on. Nothing turned up.
He had a hypothesis: Perhaps it was this synchronization that allows rhythmic patterns in muscle contraction like the wing motion of insects. It was soon disproved, however, when Elizaveta Pavlovna Chetverikova discovered the same fluctuations in an enzyme she was working with, creatine kinase, not a fibril, but a compact globular protein, not related to muscle contraction. Again the reaction rates fluctuated, and they fluctuated synchronously among her samples as well as with his ATPase reactions. Shnoll and Chetverikova then tried every protein they could get their hands on: creatine kinase, pyruvate kinase, alkaline phosphatase, lactic dehydrogenase, acetylcholinesterase, and tripsin. All showed the same synchronicity.
Could it be some fundamental property of biotic processes?
In order to make sure that what he was seeing was indeed an enzymatic reaction, and not due to some merely chemical property during the reaction, he performed what he thought would be a control experiment. He measured the reaction rate of acetic acid with dichrolophenolindophenol (DCPIP, a blue dye), a purely chemical reaction. The reaction turns the solution from blue to clear, making the rate of reaction very easy to track and making detection easy to automate. From these experiments, Shnoll found that although the amplitude of changes in reaction rate was lower, the shape of the histograms could not be distinguished from that of the enzyme reactions.
This meant that either (1) a chemical reaction was influencing the enzyme reactions, but enzymes are just more sensitive to these changes, or (2) the chemical reaction was separately undergoing the same fluctuations of rates; that is, the histograms were the same shape, although the amplitude of variation in rates (Figure 1) is process dependent.
Finding Exogenous Causes
Shnoll looked for external effects that could impact all reactions. This took many years. He and his collaborators searched for temperature, pressure, pH, mechanical, and several other possible effects. After eliminating these mechanical, thermodynamic, and chemical causes, he found that magnetic and electromagnetic fields influenced the amplitudes of both protein (e.g., enzyme) and chemical reactions.
When 25 years of data were compared to solar activity, the amplitude of scattering (the range of variation of rates) was directly correlated with the rate of change of solar activity, as measured by Wolf number (also known as the international sunspot number).
That is, regardless of the particular level of solar activity at any time, the more quickly the level of solar activity changed, the larger the jumps in enzyme or chemical reaction rate. Amplitude variation was also seen to be influenced by changes in the ionosphere (Layer F2). It was even found to be influenced by the interplanetary magnetic field, increasing two days before the Earth made a sector crossing of the interplanetary magnetic field from positive to negative. (Figure 4)
FIGURE 4

Comprehensive Solar Wind Laboratory at Goddard Space Flight Center,
http://lepmfi.gsfc.nasa.gov Shown here is the heliospheric current sheet which separates positive and negative sides of the the interplanetary magnetic field. Since it is not flat, the Earth crosses from positive to negative sides of the field many times a year. |
FIGURE 5
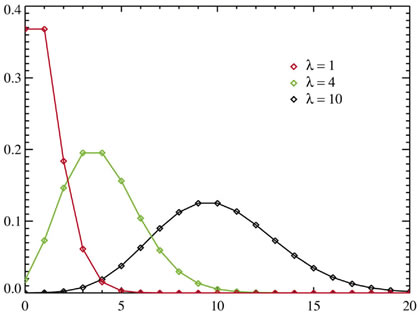
Skbkekas
Poisson distributions for a supposedly ideal stochastic process. Because the process being studied is truly random, the curves become increasingly smooth as the number of measurements increases. The x-axis shows the numbers of occurrences that can happen in the given unit of time; the y-axis shows the probability (frequency) of the different numbers of occurrences happening. |
It was clear that changes in amplitude of variation could be influenced by many factors, however the cause of the similarity of histogram shape was still not clear. To investigate the shape of the histograms, Shnoll first tried to find a completely random process to use as a control, as calibration.
Radioactive decay from one nuclear species to another, which he measured for 10 years as a doctoral student, is accepted as a completely random process in the small. But it has been a puzzle since its discovery in 1896 by Henri Becquerel, for many reasons. One is that it is both very constant and yet very erratic. Each isotope has a distinct, overall decay rate, its half-life, or the time it takes for half of the sample to decay. This rate is immutable. The only way to change it, besides dramatically changing its space-time, is by transforming it into something else, another isotope, which has a different unique half-life.
Yet, despite this predictable characteristic, the exact time at which any particular atom will undergo decay is considered to be completely random and unknowable. Any particular atom may decay right away or years hence.1 Radioactive decay, therefore, is a process that only has order statistically, macroscopically, and is therefore a truly stochastic processor so Shnoll had believedit was evident a priori.
The histogram of a truly stochastic process is a Poisson distribution. (Figure 5) The more measurements in the experiment,. the smoother the curve becomes.
Radioactivity: A Huge Surprise
Shnoll and his staff ran parallel, simultaneous measurements of carbon-14 radioactive decay in Moscow, and of the creatine kinase (enzyme) reaction in Pushchino, more than 60 miles south of Moscow, and found two astonishing results. First, the histogram of radioactive decay rates was just as differentiated, that is, unsmooth, as that for the enzyme and chemical reaction rates. Second, the two simultaneous histograms were similar.
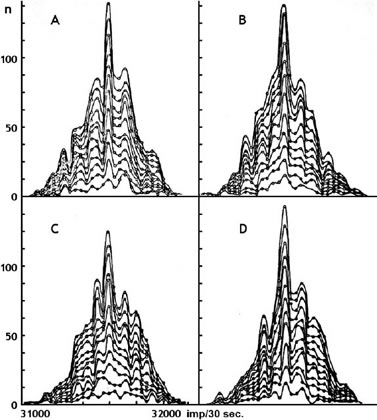
He constructed histograms from iron-55 decay data and found that increasing the number of measurements did not smooth out the curve as expected, but in fact did the opposite, making the fine structure of the curve more pronounced. (Figure 6)
Shnoll writes: The nature of the processes we studied in 1978-1985 was so diverse (biochemistry, chemistry, electricity, magnetism, beta- and alpha-radioactivity) that we could conclude: the phenomenon is independent of the type of process. And, The only common factor for all the various processes we experimented with was their occurrence in the same space-time continuum.
The Shape of Space-Time
Though Shnoll did not find the ultimate random process he was looking for in radioactive decay, he did find a great medium for his study. Radioactive decay, especially alpha decay (the emission of a helium nucleus of two protons and two neutrons), is practically independent of incidental factors. Experimenters have tried since its discovery to change its half-life, its macroscopic rate of decay, by subjecting radioactive samples to extremely high temperatures, pressures, and fields, to no avail.
What Shnoll observed did not contradict the constant nature of a nucleuss half-life. What he did see, however, is that the supposedly random, microscopic changes had a structure, and that structure was akin to that observed in so many other processes. Unlike enzymatic activity or chemical reactions, however, radioactive decay measurements are easily automated, need much less preparation, can be left alone for extended periods of time, and can be measured with high time resolution. Experiments were so easily automated that a portable version of the whole experiment was put on ships sailing to the Arctic and Antarctic, enabling the first synchronous experiments comparing results from widely separated geographical locations. Another important advantage of radioactive decay experiments is that alpha decay has a direction, and therefore spatial effects can be investigated.
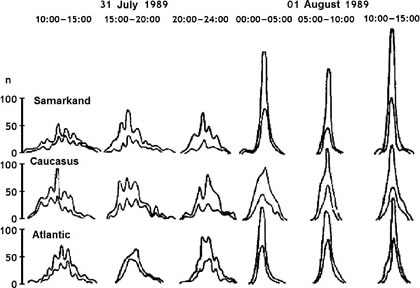
By placing radioactivity and chemical experiments on a ship traveling to the Indian Ocean, Shnolls team found that histograms recorded at Pushchino and near Madagascar6000 miles away but at nearly the same longitudewere synchronized. Results from radioactivity measurements in Lindau, Germany, however, showed histograms that were similar those at Pushchino, but offset by one hour, exactly corresponding to the difference in longitude between the two places. There seemed to some dependence on local time.
On the other hand, expeditions into the Pacific Ocean revealed another synchronicity that did not depend as much on local time and seemed to observe absolute time or a global time: that is, histograms were similar simultaneously, without time zone offset. Figure 7 shows another finding of absolutely synchronous changes in three widely separated locations.
While comparing histograms, Shnoll and his team also noticed that histograms that were one day apart were similar. In other words, a histogram may be unlike another produced from data taken minutes later, yet be strikingly similar to one taken 24 hours later.
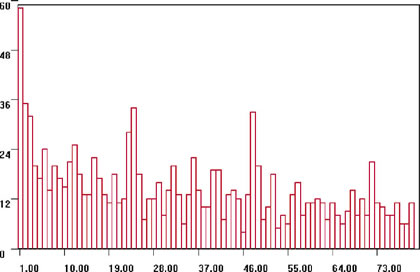
From comparing the time intervals at which histograms were similar, they made interval charts, such as Figure 8. From these time interval charts, many unexpected relationships were revealed.
Schnoll took up a suggestion that he look for similarities using the sidereal (stellar) day rather than the solar day. They differ by 4 minutes. In solar time, the moment of sunrise tomorrow is determined by a combination of the Earths rotation on its axis and its travel along its orbit around the Sun. But sidereal time is independent of Earths travel along its solar orbit.
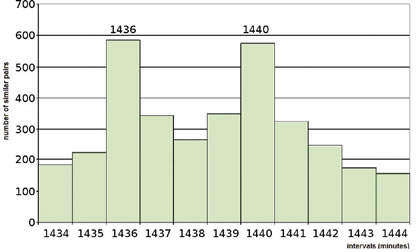
Shnoll refined his histogram intervals to minutes rather than hours, as shown in Figure 9, and compared them (he took one-second counts of decay, and made histograms of 60 different counts, amounting to one minute). When he did this, his 24-hour peak clearly separated into two peaks: one at 24 hours (1440 minutes) and one four minutes earlier (1436 minutes), exactly corresponding to the time it takes any particular place on Earth to come back to the same star (or seen inversely, for a star to come back to the same place in the sky).
From these data Shnoll concluded that, a histogram shape depends on its exposure towards the sphere of fixed stars (or the crystal canopy, as poets used to say). This moved a possible cause of the macroscopic fluctuations beyond the solar system. We got wind of inquisition fires when people inquired about these results.
Why would radioactive decay, seemingly unaffected by many extremes, be concerned about the fixed stars so far away?
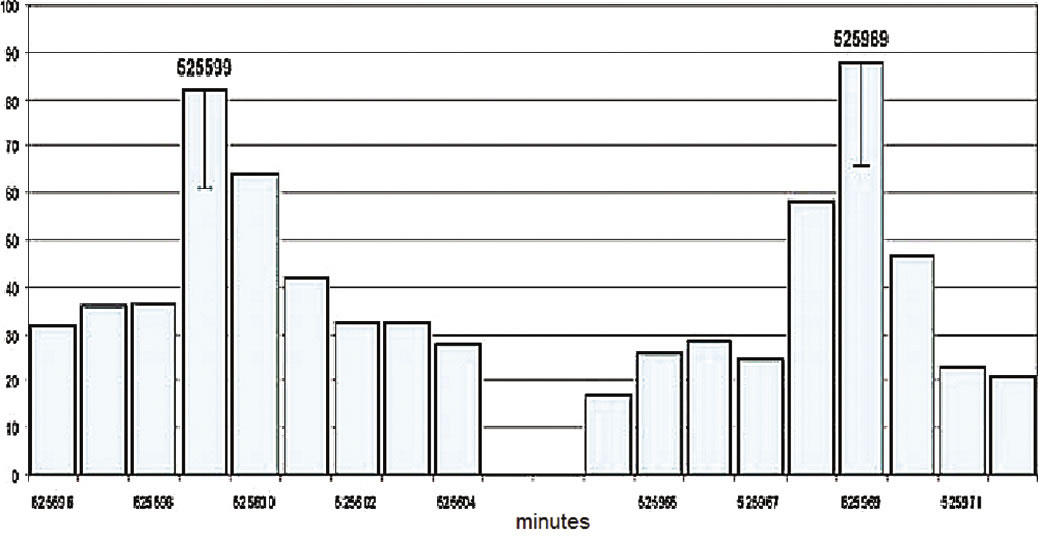
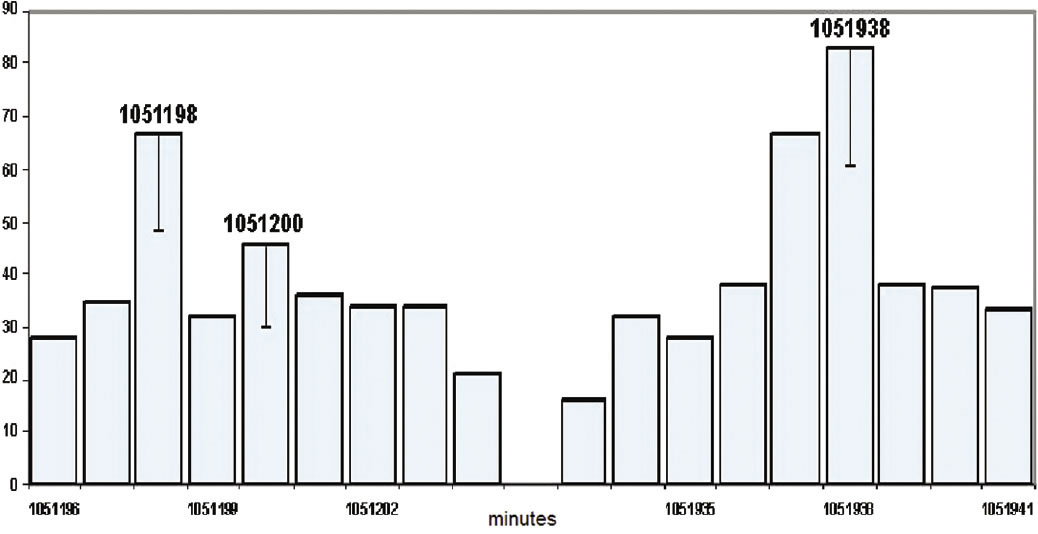
Early in his researches, Shnoll had noticed similarities in histograms of reactions of acetic acid and DCPIP one year apart, and had begun taking measurements at the same time every year. He came back to these experiments and performed them more precisely, using plutonium-239 alpha decay. Again he found that the year split into two types of years, as shown in Figure 10.
One peak occurred at 365 days (526,600 minutes), when high noon occurred 365 times per year (the calendar or solar year), while another occurred a quarter of a day later (the sidereal year). (We reconcile the two with the addition of a leap day every four years.)
There was even another peak, which was unexpected and completely unexplained. Its period was one minute less than the full calendar year. (Figure 11)
Since this cycle could not be explained by cycles within the the Solar System, Shnoll hypothesized that it may be caused by motion of the Solar System with respect to something external to it.
Again, why would decay rates or chemical reactions care about their orientation to fixed stars? Or even the Sun? Also, what would be common to all the different types of processes tested?
As mentioned, alpha decay also has the advantage of being oriented in space. The helium nucleus is not always thrown off in the same direction. In an extensive series of experiments, Shnoll and his team used collimators (directional detectors), to look at the changes in decay rate in various directionseast, west, continually toward the Sun, and toward Polaris, the star toward which Earths axis is currently pointing. From these experiments, he was able to reveal a clear anisotropy (non-homogeneity) in space. That is, direction matters. For example, certain astronomical cycles would be revealed in one direction of decay but not another.
This is just a small sampling of thousands of experiments and surprises described in Cosmophysical Factors in Stochastic Process. Every experiment was repeated many times to convince Shnolls most harsh critic, himself. He had to eliminate all possible sources of error. For all other critics, his message was, Instead of yelling, why dont you just go to the lab and repeat our experiment?2
Read the book!
More Work To Be Done
The work of Simon Shnoll and his collaborators opens up many new domains of research. The few answers they have provided only serve to stir up more questions by orders of magnitude.
First, there are the threads of research Shnoll clearly indicates would be important, but for which he has not had enough hands to continue. These include the experiments with proteins and their macroscopic conformations. What could these experiments tell us about proteins and life? Or about how the shape of space-time is reflected in other or larger life processes?
Experiments at the north and south poles, testing for effects of the solar day as compared to the sidereal day, and other effects such as the anisotropy of space, remain to be done.
Other avenues more broadly include ways to probe the shape of space-time. What features are shared by every process? Conversely, what features are uniquely expressed?
Several experiments in detecting particular directions of alpha decay prove that directions in space are not equal. Perhaps this anisotropy is due to the motion of the Earth, the Solar System, or even the Galaxy. This could be tested in a manner similar to other experiments which Shnoll performed involving collimators. Perhaps such experiments will show that anisotropy of space is not due to motion at all.
Perhaps most exciting is a sense of foray into a new physics. Shnoll had a hypothesis that perhaps gravity was at least one of the causes of variation, but experiments during times of high tides produced negative results. Hypotheses of neutrino fluxes as a cause seem to conflict with the definition of the particle as a weakly interacting one. Other hypotheses of concentrations of lepton gases also did not hold. Perhaps the effects of an entirely new, unknown principle are being detected in these experiments in that these factors do not influence the histograms, even though the histograms are changing. It may turn out that there is no simple mechanical cause, but only an overall effect of the change of space-time, as Shnoll suggests.
Can these investigations of space-time be used to probe the structure of the nucleus? Perhaps, contrary to a reductionist point of view, the very small is made of the very large.
Vladimir Vernadskys 1930 discussion of the study of life phenomena and the new physics also resonates here.3 In this paper, Vernadsky points out two gaping holes in scientific research: (1) universal principles which express themselves most distinctly in life and cognition, and (2) inherent and increasing dissymmetry of living matter.
How does the structure of space-time express itself in cognition? Can we change this structure? Life does exist in the universe. It is not an anomaly. Nor is cognition. In fact, it is these phenomena which may tell us more about the laws of the universe, since they manifest laws unexpressed in the abiotic. Can Shnolls investigation then be reversed to ask, What then is unique to the space-time of life and of cognition?
Though many questions remain unanswered, one thing is certainthere is no validity in asserting that any terrestrial process is closed, that it can be fully accounted for by terrestrial laws alone.
Footnotes
1. This randomness was used in the famous Schrödingers Cat problem, and has even been suggested to be used as a random number generator.
2. This was Shnolls rejoinder at an emotional seminar at the Moscow Institute of Physics and Technology in 1982, where Shnoll first asked for collaborators who would repeat his experiments. Cosmophysical Factors, pp. 79-80. The invitation stands: See the Shnoll Lab website at http://shnoll.ptep-online.com/index.html
3. Vladimir I. Vernadsky, 1930. The Study of Life and the New Physics.
Translated by Meghan Rouillard from lEtude de la vie et la nouvelle physique, Revue générale des sciences pures et appliquées, December 31, 1930. Washington: 21st Century Science Associates, 2015. See http://bit.ly/vernadsky-new-physics
|
|